PATH: Refitting & Repair » DIY: Propulsion » Engines » Lubrication » Oil »
PAGE CONTENTS:
Introduction / Overview.
History.
Trending.
The Value of a Single Sample Oil Analysis.
Wear Metal Analysis.
Glycol Detection, Sources, Consequences, & Removal.
Examples of Engine Damage.
Oil Analysis Labs by Regions
Documentation: Catalogs, Brochures, SpecSheets, Manuals, Parts Lists. Recalls, etc.
Forum Posts, Tech Notes & Tech Tips.
Publications & Media: Articles, Books, Magazines, Videos, Websites, Authors, etc.
Related EAB Webpages & Main Topic Pages with Links.
Visit our FEATURED ARTICLES Home Page! Thanks to our amazing contributors.
This Months Top 20 Most Popular Articles on our EAB website.
What our nonprofit Anchors Aweigh Academy and its EAB website have accomplished.
Members must SIGN IN to gain access to Members Only areas of this website.
Become an Academy Member and gain access to additional pages and programs!
Comments for everyone to view: Submit to Comments♥EverythingAboutBoats.org (Replace “♥” with “@”).
ROUGH DRAFT

For many years automotive and marine fleet operators have used engine oil analysis for two important cost saving reasons:
The first reason is to determine whither or not an engine is really in need of an oil change. Different engines wear and contaminate the oil at different rates depending on several variables such as the age of the engine, the running environment, the speed and load, and the schedule of active running verses dormancy. These variables are more thoroughly discussed in DIY: Engine Mechanical and DIY: Engine Lubrication sections.
History
+
Trending
+
The second reason automotive and marine operators use engine oil analysis also benefits aircraft operators because it offers an added safety factor by revealing internal engine problems hopefully before they suffer a catastrophic failure. An engine oil analysis can also help predict major repairs and extensive down-time for repairs. Additionally, multiple samples taken routinely from a particular engine over a period of time can be used to develop a wear trend that can predict engine life.
+
The Value of a Single Sample Oil Analysis
Private Boat owners can obviously also benefit from routine engine oil analysis for the same reasons, but can a boat owner or a perspective boat buyer benefit from a single engine oil analysis? Yes, of course! And this is how.
- A single sample, even with oil having only a few hours of running time, can reveal glycol and sodium contamination that indicates a coolant or seawater breach. If the quantities are very high, the breach is probably active. If the quantities are only moderately high (such as from carry over), then the breach was probably sometime in the past, and probably repaired. However there may still be some residual or related damage that may need to be checked out and taken care of.
- A single sample, especially with only a few hours running time can reveal elevated wear metals and contamination that indicates a poorly performed oil change that left a lot of material in the engine. This happens if the engine was not warmed up enough before the oil change, or not all the old oil was removed during the oil change. A proper oil change should leave very little contamination (called carry-over) in the engine. These engines still need a proper oil change.
- A healthy engine will produce wear metals and other contaminates at very predictable rates over the life of the oil. If the sample shows disproportionately elevated wear metals or other contaminates, this indicates that something is failing inside the engine and the contaminates abnormally elevated can indicate which part(s) are failing. All this is more thoroughly discussed later in this article.
- On occasion, a sample from an engine that reportedly had an oil change very recently, shows such proportionately elevated readings that it is difficult to believe that the oil change was actually performed. Yes, there have been boat owners that have paid someone to perform an oil change that did not really get an oil change. At the cost of quality oil and labor these days, these bills can be quite substantial, and can “earn” these unscrupulous folks a tidy bit of money. This seems to happen most often to those short season low hour recreational boat owners that conscientiously have the oil changed every year long before the oil has enough running time to become so contaminated that it needs changing. Unfortunately, the consequent of not changing the oil in these engines is sludge buildup in the oil pan during the off-season.
And yes, multiple samples that facilitates trending is of course even more valuable to a boat owner or a perspective boat buyer. If you are buying a boat, ask the owner or broker if there are any earlier oil analysis you can see. And of course, If the results are clean, they will be more then happy to share them with you.
Wear Metal Analysis
A Spectrometer such as an Atomic Absorption Spectrometer, an Inductive Coupled Plasma Spectrometer, or a Direct Current Plasma Spectrometer is used to detect wear metal particles less than five microns (a micron is one millionth of a meter). Iron, copper, lead, aluminum, chromium, molybdenum, and tin are the primary troubleshooting wear metals. It also detects silicon, sodium, and zinc.
+
Iron is a wear metal from cylinder liners, piston rings, oil pumps, crankshafts, gears, camshafts and several other parts of the valve train. Iron should not exceed 100 parts per million (ppm) in most marine engines during the normal life of the oil.
+
Some Copper normally leaches from the oil cooler core especially during periods of dormancy. A much more serious source of copper is the underlay of main bearings and rod bearings. Other sources of copper are wrist pin and rocker arm bushings, turbocharger shaft bushings, and oil pump drive. Copper should not exceed 25 ppm in most marine engines during the normal life of the oil.
+
Lead comes from the overlay on main bearings, rod bearings, and some camshaft bearings. Lead should not exceed 25 ppm in most marine engines during the normal life of the oil.
+
Aluminum comes mostly from pistons, however it also can come from the overlay of the crankshaft and camshaft bearings. Aluminum should not exceed 15 ppm in most marine engines during the normal life of the oil.
+
Chromium wears from piston rings, and ball and roller bearings. Chromium should not exceed 15 ppm in most marine engines during the normal life of the oil.
+
Molybdenum comes from the top piston ring on some engines. Molybdenum should not exceed trace amounts, however greases containing molybdenum may find their way into the oil.
+
Tin can come from the overlay on main bearings, rod bearings, and camshaft bearings. Tin should not exceed 15 ppm in most marine engines during the normal life of the oil.
+
Silicon in a special form is used as an oil additive to retard foaming of the oil. Silicon in the form of “dirt” should not exceed 15 ppm in most engines during the normal life of the oil.
+
Sodium in a special form is used as an oil additive. Sodium as a salt is found in antifreeze and salt water and may indicate a coolant leak or wet exhaust system problem such as a breached exhaust elbow.
+
Zinc is an oil additive and often exceeds 1000 ppm in better oils.
+
Oil Condition Analysis
+
Wear often occurs when the oil itself has deteriorated due to depletion of its additives or has become contamination from outside sources. An instrument called a Fourier Transform-Infrared Analyzer focuses a beam of light through a film of used oil and compares the light transmitted at certain wavelengths to those of the same oil when new. To receive the greatest benefit from this test, a sample of unused oil saved from the same batch as was drawn from the engine is sent to the lab to be used as a reference in the analysis. Soot, oxidation, nitration, and sulfonation can be detected with the FT-IR instrument.
+
The rate that soot (partially burned fuel) accumulates in the oil indicates the quality of combustion in the engine. A rich fuel to air mixture, low compression, low running temperature, or fouled injector(s) will accelerate the accumulation of soot in the oil. Excessive soot degrades and thickens the oil and will lead to bearing damage as well as accelerated piston, ring and liner wear.
+
Oxidation and nitration normally increase at a steady rate throughout the life of the oil. Excessive oxidation or nitration may indicate that the optimum drain interval has been exceeded. If the oil is still fairly new, it may indicate higher than recommended operating temperatures or overheating. Oxidation and nitration of the oil will increase its viscosity and leads to plugged filters, lacquering, piston deposits, ring sticking, and bore polishing.
+
Sulfonation relates to the oil’s alkalinity reserves (measured by Total Base Number) and the oil’s acidic property (Total Acid Number). The TBN indicates whether enough of these additives remain to protect the engine from acid formation, and the TAN indicates the relative level of acid formation. Rapid alkalinity reserve depletion and acid formation can result from high air humidity, low running temperature, and/or high idle time. Corrosive acids formed during engine operation can etch metal parts causing rapid wear.
+
Contamination
The FT-IR instrument also scans for the presents of water, glycol (antifreeze), and fuel. If the possibility of water, glycol, or fuel is indicated, a separate physical test is performed for verification.
+
Water sometimes contaminates engine oil through a leaking gasket or seal. However, low running temperature can allow condensation of combustion blow-by gases (mostly H2O steam that passes from the combustion chamber past the piston into the crankcase). Exhaust elbows that are to low to the waterline in wet exhaust systems and water lift muffler equipped systems lacking an anti-siphon vacuum break are far too often a source of water contamination.
+
Glycol contamination of the oil indicates an antifreeze coolant leak. Pressure testing the cooling system may reveal the location of the leak.
+
Fuel contamination of the oil indicates a fuel system problem such as a leaking fuel injector(s) or a ruptured diaphragm in the fuel supply pump. Fuel dilutes the oil reducing its viscosity and lubricity resulting in bearing damage, and accelerated piston, ring and liner wear.
+
To get the greatest benefit from oil analysis
1. Provide all the information about the engine as requested on the sampling label. The make and model of engine, the hour meter reading, hours on the oil, and the brand and weight of the oil are critical for a complete analysis. The date the sample was taken is also important. The serial number helps determine the vintage of the engine and identify the engine for trending with later sampling. Was any make-up oil added that would affect the analysis? Were the filter and/or oil changed at time of sampling? Don’t forget to include the vessel identity and contact information. If the vessel has more than one engine, identify which engine the sample is from. Filling out the label before taking the sample may help keep the label clean.
2. Take oil samples at regular intervals. As the number of samplings increase, chart the results to find trends that would indicate trouble.
3. Use oil analysis to help determine the most cost-effective oil change interval for this particular engine.
4. Use oil analysis to discover problems and than confirm the source(s) of the problem through further testing and inspection before they result in major damage or failure. Exhaust smoke, fuel consumption, loss of power and internal noises can help to establish engine problems. Oil analysis should not have to be used as the sole indicator of trouble.
+
Other Fluid Analysis
+
Engine coolant analysis can be quite helpful in discovering engine problems such as a leaking oil cooler, an air leak into the coolant, defects in electrical ground connections, spent metal pitting protection, leaking head gaskets, and destruction of iron, copper and aluminum components.
+
Marine gear fluid analysis can detect impending failure from several sources including overheat, excessive wear, and water intrusion.
+
Hydraulic fluid analysis helps discover system wear and possible damage.
+
Diesel fuel analysis can reveal a microorganism infestation and other contaminates.
+
Many laboratories offer all these analysis, and generously provide help to those needing these services. I have found the Caterpillar dealerships that operate Cat’s S-O-S program to be especially helpful.
+
Glycol Detection, Sources, Consequences, & Removal.
Glycol In Lubricating Oil – Detection, Analysis and Removal
Tags: contamination control
Introducing glycol into lubricating oils exposes your machines to a powerful and poisonous mixture of chemicals. Unlike other harmful contaminants such as water and dirt, the destructive potential of glycol can progress to massive failure of machine components in a narrow window of time.
There could hardly be a more important role for the oil analyst than the routine screening of lubricating oils for the presence of glycol. One large oil analysis laboratory that specializes in heavy-duty fleet equipment deployed in mining and construction reported that glycol was found in 8.6 percent of motor oil samples over a period of years – about one in 12 samples.
In fact, oil analysis laboratories have been testing for coolant leaks in used motor oils and other drive train lubes for nearly as long as used oil testing has been around. The techniques vary considerably from lab to lab, as do the alarming limits. This article brings the subject of glycol testing in the oil analysis lab into the spotlight by describing the latest laboratory techniques for detecting and measuring its concentration plus tips for troubleshooting.
How Glycol is Formulated and Used
Glycol, the principle ingredient in antifreeze, is typically mixed with water 50/50 to form a liquid “coolant” to transfer heat, raise boiling point temperature (above 225°F or 107°C) and lower the freezing temperature (below -32°F or -35°C). When additives are incorporated into the formulation, thecoolant can guard effectively against corrosion and cavitation.
Both propylene glycol and ethylene glycol are used in coolant formulations. Propylene glycol is the choice of some users, because unlike ethylene glycol, it is neither toxic nor considered a hazardous material. Ethylene glycol is far more widely used, however, primarily due to its higher heat transfer properties. This article will focus entirely on ethylene glycol.
Antifreeze formulations used as coolants deploy an assortment of organo-metallic and organic additives. These are used to protect metals in the cooling system from corrosion/cavitation, to control scale, to prevent foaming and to maintain pH. Common examples of additives include various phosphates, sodium borate, molybdate, sodium silicate, potassium sebacate and sodium nitrate.
Just like additives in a lubricating oil, these additives will contribute to ranging elemental concentrations of sodium, boron, potassium, silicon and phosphorous in the coolant. As will be discussed in greater detail later, the elemental families from the coolant additive system help serve as markers, like DNA, to identify glycol contamination of lubricating oils.
Additive treatments used in antifreeze formulations vary considerably between aftermarket suppliers and OEMs that provide the original factory fill and offer supplemental coolant additives (SCAs). There are also marked geographical differences in coolant additive chemistry, influenced by environmental regulations and water quality. For instance, the Japanese use no silicates but large amounts of phosphates.
Conversely, the Europeans deploy silicates, benzoate, borate, nitrite and nitrate additive compounds. U.S. formulations employ silicates, phosphates, as well as numerous organic inhibitors.
How Glycol Gets into Motor Oils and Other Lubricants
Glycol can leak into motor oils and other lubricating oils in a variety of ways. These include:
- Defective or deteriorated seals
- Blown head gaskets
- Improperly torqued head bolts
- Thermally warped or cracked cylinder heads (from low coolant charge to stuck thermostat)
- Cracked block or cylinder head from frozen coolant
- Improperly machined head and block surfaces
- Corrosion damage of cylinder liners
- Cavitation erosion/corrosion of cylinder liners
- Electrochemical erosion
- Damage or corroded cooler cores
- Water pump seal failure and weep-hole blockage
In fact, a major diesel engine OEM has estimated 53 percent of all catastrophic engine failures are due to coolant leaks. For many diesel and natural gas engines, the highest risk of contamination occurs during the time the engine is not operating. In such instances, the cooling of the engine from intermittent use can lead to internal leaks associated with thermal creep, such as at the cylinder heads where there is risk of recession or movement from gasket seals.
Higher hydrostatic pressures of the coolant in relation to the lube oil system compound the risk when the engine is at rest. This can lead to a slug feeding of the coolant into the lubricant.
Cylinder Liner Cavitation
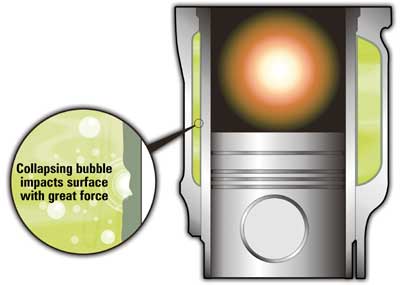
Another common source of leakage in engines with wet cylinder liners is associated with a chemi-mechanical perforation of the liners, promoted by vaporous cavitation. This occurs when the liners vibrate violently (on the load side) to the rhythm of piston movement, compression and combustion. This movement causes the rarefaction portion of pressure waves to form negative pressure regions that nucleate vapor bubbles (cavities).
As the combustion chamber fires, the vapor bubbles implode at the speed of sound causing fluid jets and surface pressures as high as 60,000 psi. Such localized energy can literally blast small holes in the protective oxide film on the liner wall, similar to vaporous cavitation in hydraulic pumps.
The damage can be further propelled by chemical attack of the nascent metal exposed during this cavitation event. Over time this can lead to perforation of the liner and leakage (Figures 1 and 2). Note, many researchers have postulated on the exact mechanism of damage. While there are variations in theory, there is general agreement that the failure mode is propagated by a combination of mechanical (localized cavitation) and chemical (corrosion of the exposed base metal) attack.

Cylinder Wall Cavitation Erosion
Certain additives used in SCAs, such as molybdate and sodium nitrite, have been found to sharply inhibit the progress of cavitation corrosion. If the liner’s protective oxide film becomes delaminated by the cavitation energy, the additive reforms a barrier film to stop further progress.
The concentration of these SCAs introduced to the coolant blend is important however. An undercharge can result in accelerated pitting while an overcharge can cause coolant gelation, lead-based solder corrosion and other problems.
Harm Caused by Glycol-Contaminated Lube Oil
Glycol is an all-around bad guy when it commingles with oil. The problem is intensified by coolant water that enters the lube system at the same time as glycol. Evidence of glycol contamination is often seen by mechanics charged with the responsibility of repairing the damage it causes.
For instance, main and connecting rod bearings may become darkened, almost charcoal in appearance, when glycol has contaminated diesel engine crankcase lubes.
Owing to the fact that glycol is not soluble in mineral oil and that the thermal conditions within engines lead to the transformation of the glycol and coolant additives to an assortment of other chemicals, it is no surprise that there are so many degenerative consequences.
Following is a discussion of several common and a few not-so-common symptoms or harmful effects of glycol leakage and contamination.
Hydraulic Lock
It was previously mentioned that coolants could corrode and erode the walls of cylinder liners. This can result in pinhole perforations. When an engine is at rest, the combustion chamber within the cylinder can literally be flooded with coolant passing through these holes. Later, when cranking the engine, the lack of compressibility of the coolant can cause hydraulic lock, like deadheading a pump. Since the coolant has nowhere to go, bearings, rings and/or rod failure can occur.
Acid Formation and Bearing Damage
Under normal operating conditions, ethylene glycol oxidizes to form organic acids such as glycolic acid, oxalic acid, formic acid and carbonic acid. Typically the reaction rate doubles for each 18°F (8°C) in temperature. These acids contribute to secondary and tertiary effects as further described. However, their presence alone in lubricating oil can jeopardize bearings and other frictional surfaces.
The corrosive conditions can pit the clad surfaces of the lead/tin overlay of journal bearings, promote rust on steel and iron surfaces, and tarnish cupric metals of bronze and brass. One study found that even a small coolant leak in a large integral gas engine/compressor was enough to severely corrode engine steel and copper surfaces.
|
Figure 4. Additive Precipitation – 2 Percent of a 50/50 Ethylene Glycol Water Solution Added
|
|
|
Oil
|
Filterable Solids
(Grams per Gallon) (3.79 Liters) |
|
A
|
77
|
|
B
|
40
|
|
C
|
33
|
|
D
|
26
|
|
E
|
17
|
|
F
|
10
|
|
G
|
8
|
|
H
|
4
|
|
I
|
1.6
|
Oil Balls and Additive Precipitation
Chevron and other researchers have reported that when glycol-based coolants thermally age in crankcase lubricants oil balls form, largely from the reaction of the glycol with oil additives. The additives involved include sulfonates, phenates and ZDDP (Figure 3). This is further evidenced in a study by the Fleetguard filter division of Cummins Engine that reported as much as 77 grams of filterable solids are formed when oil is contaminated with coolant containing ethylene glycol at a concentration of just two percent (Figure 4).
Loss of Dispersancy and Filter Plugging
The acids and water that form in crankcase oil as a result of coolant contamination will often disrupt soot dispersancy, even at low soot loading. Fleetguard reports “75 percent of filter plugging complaints from customers involved coolant or moisture in the crankcase.” Once soot begins to dump, a chain reaction of associated failures can result, including loss of antiwear protection, sticky sludge on valve deck surfaces and carbonaceous deposits on ring grooves, piston crown lands, valve train components and oil ways to bearings, etc.
If the problem is not identified, the oil is often changed without a system flush (described later in the Procedure for Cleaning Glycol from Engines section). The chain reaction then gains new life as the detergents and dispersants coming in with the new motor oil can mobilize the sludge and deposits. Then, within minutes after an oil and filter change, the new filter can become plugged again. Following is the summary of this chain reaction:
1. Coolant leaks into the crankcase oil.
2. Acids and precipitants form as the glycol, coolant additives and lubricant additives react.
3. These insolubles begin to plug the oil filter.
4. Concurrently, the acids and water disrupt soot dispersancy causing a dumping condition. More sludge and insolubles form.
5. By now the filter is plugged with the glycol transformation by-products and coagulated soot.
6. The oil and filter are changed (typically around 15 percent of the old oil remains, either in the oil pan or occluding to engine surfaces). The new oil (with detergents and dispersants) mobilizes the soot and the sludge, carrying it to the filter.
7. Once again, the filter becomes plugged (even with the coolant leak fixed).
Oxidation and Viscosity Change
When glycol contaminates lubricating oils, the viscosity of the oil can increase dramatically. This problem is especially acute in highly additized motor oils. High viscosity can lead to inadequate flow of the lubricant to critical frictional surfaces. So too, glycol and its reaction products can aggressively promote oxidation of the base oil. Caterpillar reports that “coolant contamination in transmissions and hydraulic fluids usually shows up as an increase in oxidation.”
How Glycol is Detected in the Field
Astute mechanics, lubrication technicians and operators are always on the lookout for the telltale signs of a lubricant contaminated with coolant. In truck, bus and mobile equipment fleets, the first sign might be white smoke billowing from diesel engine exhaust pipes. Or it might appear as a shiny adherent sludge having the consistency of mayonnaise on a used filter during a routine change out. Perhaps as previously mentioned, the oil pressure in a diesel engine runs unusually high only minutes after an oil and filter change.
Blotter Spot Test
One test that has gained renewed attention is the blotter spot test. It first appeared on the lubrication scene as a field test dating to about 1880. It reemerged in studies conducted by Shell Oil in the 1950s and now again it seems to be catching the attention of even the most sophisticated oil labs. Because of its simplicity, the test is easily conducted in the field, although time is required for the results to fully develop.
The test is based on the established procedure of paper chromatography and involves placing a couple of drops of used oil on common blotter paper (available from lab supply catalogs), or even the back of a business card. Let the drops soak into the paper for a couple of hours. If a dark or brownish stain is left in the center after the oil absorbs outward, then this could be disrupted dispersancy and soot coagulation, a common consequence of glycol contamination.
A black sticky paste with a well-defined (sharp edge) periphery is cause for serious concern. Very often a soot ring develops around a yellow/brown center when glycol is present. Figure 5 shows this unique pattern on blotters developed from used diesel crankcase oil that was thermally aged in the presence of varying concentrations of glycol and water.
Patch Test
When passing a small amount of solvent-diluted oil through a one-micron membrane you will often see sludge, additive precipitants and other insoluble contaminants present on the membrane surface. The use of a handheld 30-power microscope can help in the examination of the material present. Field patch test kits are available from several suppliers.
Note if the membrane pore size is too large (>3 microns), much of the sludge and insolubles will pass through. The use of a 0.8 mm patch is recommended. Also avoid the use of solvents that risk dissolving condensed oxides and other target materials from glycol-contaminated oil.
Schiff’s Reagent Method
The Schiff’s reagent method (ASTM D2982) is a colorimetric method for detecting trace amounts of glycol in lubricating oils. In this method, a solution of hydrochloric (HCl) and periodic (HIO3) acids is introduced to the oil to oxidize any glycol that may be present. The reaction produces an aldehyde, which in turn reacts with the Schiff’s reagent, yielding a positive color change from colorless to pink/purple – the darker the color, the more glycol present. There are several suppliers of Schiff’s reagent test kits.
Please note, false positives have been frequently reported from the presence of trace impurities in new oils. Chevron has documented this effect as being due to residual traces of glycol in new oil from lubricant additive manufacture. However, others have reported that it might also be due to aldehydes and ketones from base oil refining, despite the published Chevron data. This makes sense because new oil can show a positive with Schiff’s before oxidizing with HIO3.
In another case, the Cummins oil analysis lab reported that it ran successive tests on a single sample of used oil that yielded results that were positive, negative and indeterminate. Because of the instability of ethylene glycol in lubricating oils at high sump temperatures, it is rapidly converted to acids and other compounds. Because this and other test methods discussed below rely on the presence of molecular glycol, any chemical or thermal decomposition of the coolant can make the Schiff’s reagent test unreliable. Lab tests by Cummins show that “an oil containing four percent coolant will retain only 10 percent of the glycol originally present upon heating at 200°F (93°C) for eight hours.” However, other telltale signs for glycol contamination do remain in the oil.
How Glycol is Detected in the Lab
Commercial oil analysis labs deploy a number of different test methods and instruments to detect and measure glycol concentrations in used lubricants. The effectiveness of these techniques varies considerably, and for this reason, a discussion that compares and contrasts them is warranted. To this end, the three most common methods used by oil analysis labs are discussed below:
Fourier Transform Infrared Spectroscopy (FTIR)
Many oil analysis laboratories use this procedure for analyzing used oil molecular properties routinely. Practicing Oil Analysis magazine has covered infrared spectroscopy in several issues. Common target properties detected and measured with FTIR include soot load, water contamination, fuel dilution, oxidation, nitration and sulphation. Many labs also report absorption bands commonly associated with ethylene glycol.
While there is much merit to including glycol among the various properties reported by infrared, there are also interferences and lower sensitivity limits that labs and users need to be aware of. The interferences relate to other properties and contaminants that share common spectral absorption regions with glycol.
For instance, the strongest band for glycol, a broad region around 3450 cm-1 corresponding to the O-H functional group, is also cohabited by water and other alcohols that have similar functionality. Because water is blended with antifreeze (ethylene glycol) to form coolant, the distinction is often impossible to make.
There is however, a more unique band for ethylene glycol at 1070-1030 cm-1 corresponding to the C-O functional group, that is commonly used because there are fewer overlapping bands from other oil contaminants and degradation by-products (Figure 6). The main risk in this region is associated with certain additives (such as barium sulfonate) and oxygenated compounds (ethers, lactones and alcohols) from base oil and additive degradation.
As with water contamination, there is very limited ability using FTIR to detecting glycol concentration below 1,000 ppm. Just like the Schiff’s reagent test, FTIR also relies on the presence of molecular glycol. However, because glycol can rapidly degrade chemically and thermally as previously mentioned, its presence in used oil may be completely disguised. In this instance, for FTIR to be successful, an astute analyst would need to recognize the absorption bands of glycol’s several transformation products.
Gas Chromatography
There has perhaps been no analytical method that has had a greater impact on petroleum analysis than gas chromatography (GC). In the realm of used oil analysis, it is widely held to be the most accurate method for detecting and measuring glycol and fuel, although more costly and time-consuming than alternatives. The most commonly used GC procedure for glycol analysis is ASTM 4291, “Standard Test Method for Trace Ethylene Glycol in Used Engine Oil” (Figure 7).
The procedure involves first extracting the glycol from the oil using water followed by centrifugation. The extract is then removed and injected into the GC where the polar compounds are separated and detected on a chromatogram.
Note, this procedure has been reported by Chevron to be so sensitive that it can give false coolant leak indications. This is due to the presence of additive manufacturing by-products and fuel contamination that can leave trace levels of glycol in new and use oils. It is helpful to baseline new lubricants using GC before deploying the method with used oils.
The major drawback with GC detection of glycol is that glycol can rapidly decompose into acids, aldehydes and esters which can result in a false negative or understatement of the true concentration of glycol and glycol derivatives. This is also true for Schiff’s reagent and FTIR tests.
Elemental Analysis of Used Oil and Filters
Elemental analysis by Inductive Couple Plasma (ICP) emission spectroscopy or by arc-spark Rotating Disc Electrode (RDE) emission spectroscopy has been the mainstay of the oil analysis lab for years.
The use of elemental analysis is perhaps the single most reliable method to verify that glycol or its derivatives have contaminated a lubricant. The procedure is keyed to detecting the presence of organo-metallic antifreeze additives that become blended with a lubricant on contamination. These elemental markers are telltale signs that the oil has received a dose of antifreeze.
For instance, sodium, boron, potassium and silicon are commonly found in antifreeze formulations (Figure 8). In order to know specifically which additive elements and their concentrations are found in new or used antifreeze (included blends containing SCAs), a sample could easily be analyzed in the same way as used oils.
Of the different compounds that make up antifreeze additives, sodium and potassium compounds are the most stable, even though there is risk that they will precipitate and be removed by the filter. Boron presents a risk of evaporating at typical crankcase sump temperatures, while silicon from a coolant leak may be confused with dirt ingression or oil antifoam additive. See the sidebar for developing a calibration curve for glycol contaminant levels using sodium against various antifreeze concentrations.
Quoting again from the report by the Fleetguard division of Cummins Engine, “Oil samples will sometimes have several hundreds parts per million sodium, yet there will be no moisture or glycol present. The amount of sodium indicates that at least a gallon of coolant has leaked into the crankcase, but there is no sign of it (other than the sodium).” Cummins summarizes by saying, “Our experience is that the most reliable indicator of coolant leakage is the sodium level of the filter paper ash followed by the level of the sodium in the oil.”
Due to the loss of sodium from oil consumption or by insoluble sodium captured by the oil filter (80 to 90 percent of the total sodium that has leaked into the crankcase in some instances), an increase in sodium in the oil by as little as 50 ppm can mean as much as one gallon of coolant has leaked into a 10 gallon (38 L) lube oil system.
If the residue of a used filter is suspected to be coolant additives, an analysis to determine sodium could be performed as follows:
1. Using an ultrasonic bath, transfer the particulates from a piece of the filter media to a solvent such as kerosene or mineral spirits.
2. Use an acid digestion method to determine the additive elements (sodium, etc.) in the filter residue, or
3. Transfer the residue to a filter membrane and analyze the materials using x-ray fluorescence spectroscopy (or SEM/EDX,
ΞPIXΞ
E), or
4. Transfer the residue to a glass slide and analyze the particulate matter using chemical microscopy.
When a sudden increase in sodium is observed, the analyst needs to be aware of other common sources. These include: salt and saltwater, grease, dirt, fly ash, sodium hydroxide, etc. For this reason, other members of the coolant additive family may be needed to confirm the contaminant is antifreeze, such as boron, potassium, silicon and phosphorous, bearing in mind that some of these elements may also be oil additives.
Limits and Alarms
Setting alarms and limits for glycol is difficult because of the different oil analysis instruments in use and the transient states of glycol in the lubricant. To the extent that the analyst can confirm a coolant leak, no matter how small, the matter is of serious concern. However, false positives can occur if the alarm limits are set too low for the reasons previously discussed. Likewise, if the alarms are set too high, the actual coolant leakage levels could be several times higher than the measured amounts and the engine may be dangerously close to catastrophic failure.
In the case where false positives occur as a result of new oil chemistry associated with additive manufacturing and base oil refining, the interferences that cause these false positives often burn off during service. Hence a new reference oil or an oil placed into service a short period of time may test positive for glycol, then later test negative.
When a false positive due to oil chemistry occurs in labs that report the result of a glycol test (Schiff’s reagent, FTIR and GC) they are typically bound to place a positive indication on the report. However, based on their review of other indicators (including coolant additive elements) as previously discussed, the analyst commenting on the oil analysis data should emphasize that the positive glycol result is not a cause for concern.
Some labs use the approach of reporting coolant or antifreeze contamination instead of glycol to eliminate this potential cause for unwarranted concern. The same principle holds true in the case of a negative glycol test where its presence is disguised by the fact that it has converted to by-products.
Looking at other indicators such as sodium and potassium, the analyst should report a positive for coolant contamination despite the fact that no actual glycol was detected. In such instances, the lab analyst is making a contamination entry determination based on review of multiple factors.
In general, glycol above 200 ppm in most cases is considered reportable. Levels greater than 400 ppm should be regarded as significant and levels as high as 1,000 ppm flagged as critical.
Procedure for Cleaning Glycol from Engines
A flushing procedure proposed by one major oil company to remove glycol from engine internal surfaces involves the use of Butoxy-Ethanol (trade name Butyl Cellosolve, Union Carbide). The flushing procedure for conditions of less than five percent glycol contamination is summarized below:
1. Drain oil from compartment and all lines and components.
2. Install new filter elements.
3. Mix an ISO VG 32 R&O mineral oil 50/50 with Butyl Cellosolve.
4. Use the mixture to turbulently flush (using an external pump installed at the turbocharger supply line or other suitable pumping method) internal surfaces by ample flow and volume for one hour at a temperature not less than 70°F (21°C).
5. Drain the system completely, mop out crankcase and replace filters.
6. Repeat steps 2 through 5, using a 60 percent R&O 32, 40 percent kerosene mixture.
7. Remove and inspect all main bearings, journals and other engine surfaces.
8. Remove the external pump and recharge with the correct lubricant. Replace the filter.
9. Monitor the engine carefully until all conditions stabilize.
References:
Anderson, Daniel P., Lucas, Malte, and Lynch, Brian K. – Spectro Incorporated. “Diesel Engine Coolant Analysis.”
Anonymous (Phillips 66). Cleaning Procedures to be Used When Lube Oil Systems are Contaminated with Ethylene Glycol.
Annual Book of ASTM Standards, 2000, Section Five, Petroleum Products, Lubricants, and Fossil Fuels.
Beal, Roy E. (1993). Engine Coolant Testing, Third Volume, ASTM.
Birke, Mike – Southwest Research Institute (1999). “New Challenges in Detecting Glycol Contamination with New Anti-Freeze Additive Formulations,” Practicing Oil Analysis ‘99 Conference Proceedings.
Booser, Richard E. (1997). Tribology Data Handbook, CRC Press.
Caterpillar, various S·O·SSM Services publications.
Chevron Technical Bulletin LTB-03 (1993). “Avoiding False Coolant Leak Indications with Lubricating Oil Analysis.”
Coates, J.P., and Setti, L.C. (1985). Oils, Lubricants, and Petroleum Products, Characterization and Infrared Spectra. DEKKER.
Hudgens, R. D., and Feldhaus, L. B. – Fleetguard, Inc., (1978). “Diesel Engine Lube Filter Life Related to Oil Chemistry,” International Fuels & Lubricants Meeting, Society of Automotive Engineers, Toronto.
Mathys, Mark – Butler Machinery, Caterpillar (1999). “Coolant and Coolant Analysis,” Seminar Course Materials.
Peterson, F.S. (1979). Lubrication – Chromatography. Texaco, Vol. 65.
Staff editor. (1999). “Schiff’s Reagent Glycol Test – Controlling False Positives,” Practicing Oil Analysis magazine, November-December.
+
Examples of Engine Damage
ΞPIXΞ
s

Very high aluminum levels in the oil helped determine how badly this engine was overheat damaged when the raw water pump drive belt broke.
 High lead and tin levels in the oil showed the extent of the damage to the main bearings after this engine ran low on oil when the hose to the oil cooler failed after chaffing on the oil filter bracket.
High lead and tin levels in the oil showed the extent of the damage to the main bearings after this engine ran low on oil when the hose to the oil cooler failed after chaffing on the oil filter bracket.
 High iron, lead, tin and oxidation levels predicted the debris damage to this main bearing and its cause. Several camshaft lobes distempered from friction and wore flat because the oil broke down after a worn-down raw water pump impeller cam caused the engine to ran at an elevated temperature.
High iron, lead, tin and oxidation levels predicted the debris damage to this main bearing and its cause. Several camshaft lobes distempered from friction and wore flat because the oil broke down after a worn-down raw water pump impeller cam caused the engine to ran at an elevated temperature.
 The engine containing the top main bearing tested positive for water and showed elevated lead and tin indicating excessive bearing wear. If it hadn’t been caught and the source of water repaired, this bearing would have worn until it looked like the bottom bearing. The engine containing the bottom main bearing was showing elevated copper after the overlay was worn away exposing the copper. If this problem had not been discovered, the prospective buyer would have unknowingly purchased a vessel needing extensive repair.
The engine containing the top main bearing tested positive for water and showed elevated lead and tin indicating excessive bearing wear. If it hadn’t been caught and the source of water repaired, this bearing would have worn until it looked like the bottom bearing. The engine containing the bottom main bearing was showing elevated copper after the overlay was worn away exposing the copper. If this problem had not been discovered, the prospective buyer would have unknowingly purchased a vessel needing extensive repair.
 Elevated iron, lead, tin and copper helped discover the bent rod that this engine suffered when water intruded through a poorly installed exhaust system and hydraulic-locked the piston when it was started.
Elevated iron, lead, tin and copper helped discover the bent rod that this engine suffered when water intruded through a poorly installed exhaust system and hydraulic-locked the piston when it was started.

Note the wear pattern into the copper layer of the bearing. This is due to the rod being bent and is called radius ride.


Oil that tested positive for water, glycol and high levels of sodium revealed the perforation of this cylinder wet liner from corrosive pitting that allowed coolant to leak into the crankcase.
+
Reading results of engine oil analysis:
Aluminum: should be 15 PPM or less. Usually comes from pistons, maybe crankshaft or camshaft bearings.
Chromium: should be 15 PPM or less. Usually comes from piston rings, and ball/roller
bearings.
Copper: should be 25 PPM or less. Usually comes from oil cooler.
Iron: should be 100 PPM or less. Usually comes from cylinder liners, piston rings, oil pump.
Lead: should be 25 PPM or less. Usually comes from main, rod or camshaft bearings.
Molybdenum: should be trace amounts. Usually comes from top piston ring.
Silicon: should be 15 PPM or less, unless oil contains it as an additive.
Sodium: indicates coolant or wet exhaust leak, unless oil contains it as an additive.
Tin: should be 15 PPM or less. Usually comes from main, rod or camshaft bearings.
Zinc: common oil additive; better oil may have 1000 PPM or more.
Tips:
Rate of soot accumulation in oil indicates quality of combustion. More soot means bad
combustion, and degrades oil effectiveness.
Oxidation and nitration: increase over life of the oil. If high, caused by too long between
oil changes, or overheating.
Sulfonation: low TBN and high TAN mean too acidic, from low operating temperature or lots of idling.
Water: could be from low operating temperature, as well as a leak.
Also can test coolant, hydraulic fluid, transmission fluid.
+
Taking the Baseline Sample
An oil sample taken immediately after an oil change can serve as a
baseline or reference point for comparison with later samples at
regularly scheduled maintenance intervals (250 hours, 500 hours,
etc.). The baseline oil sample enables you to determine the amount of
contaminant carryover.
Taking a baseline sample is easy and doesn’t require additional
downtime of the machine. Run the engine for about 15 minutes before
taking the sample.
+
Oil Analysis Labs by Regions
Blackstone Laboratories (Fort Wayne, IN)
Caterpillar S-O-S.
^ NC Machinery (Tukwila, WA)
Chevron LubeWatch (US)
^ Texaco LubeWatch (Europe)
Polaris Laboratories (Indianapolis, IN)
Spectra Labs (Tacoma, WA)
+
Product Documentation
Forum Posts, Tech Notes & Tech Tips
| TYPE: | |
|---|---|
| TITLE (NOTES) — AUTHOR. | |
| Forum Posts: | |
| + | |
| Tech Notes: | |
| + | |
| Tech Tips: | |
| Tech Tip #14 Using Oil Analysis to Avoid Downtime – Part 1 from FoleyEngines.com | |
| Tech Tip #18 Oil Analysis Procedures from FoleyEngines.com | |
| + | |
| Tech Tip ## from FoleyEngines.com | |
| + | |
If you think we should add a Forum Post, Tech Note or Tech Tip to this section, please submit the Link via email To: Editor♥EverythingAboutBoats.org (Replace "♥" with "@")
Media
Related Media
with Links to EAB's Media Overview and Creator Profile Pages
Media with Bold Titles are part of our Academy eLibrary!
Academy Members can view the Media by clicking on its Bold Title Link to go to its
EAB overview page and then scrolling down to the "Academy eLibrary" section for its link.
To help us alphabetize the media lists below, each leading grammatical
article ("The" - "A" - "An") has been moved to the end of the title.
DS = Data Source code
| TYPE: | |
|---|---|
| TITLE — AUTHORS‚ EDITORS‚ PUBLISHERS‚ PRODUCERS‚ DIRECTORS‚ ACTORS‚ etc. | |
| Articles: | |
| A Pair of Very Interesting Articles on Oil Analysis from SAMS® Spring 2001 Newsletter. | |
| Glycol In Lubricating Oil – Detection‚ Analysis and Removal — Machinery Lubrication | |
| + | |
| Books: | |
| + | |
| Magazines: | |
| ANCHOR‚ The — Anchors Aweigh Academy. | |
| DIY Boat Owner – The Marine Maintenance Magazine | |
| ^ Troubleshooting with Oil Analysis – Issue 2003-3 – Page 20 | |
| Videos: | |
| + | |
| Websites: | |
| + | |
If any Media or Creators should be added to this list, please submit their info/links via email To:
Editor♥EverythingAboutBoats.org (Replace "♥" with "@")
NOT AN ACADEMY MEMBER?
CLICK HERE to discover how you can become a Member and gain FULL access to
thousands of expanded pages and dozens of excellent programs including our eLibrary!
CLICK HERE to view ALL the books, magazines, videos, etc. in our Academy eLibrary.
Media are also listed by category on the Topic Pages found on the Right Sidebar ⇒
CLICK HERE to donate any books, magazines, manuals, or videos, etc. to our Library.
Related EAB Webpages
Taking a Proper Engine Oil Sample for Analysis.
Microscopic examination of an oil filter element
+
02 – BOAT BUILDING, OUTFITTING, REFITTING & REPAIR: (Incl. DIY).
02.01 – Boat Designing Schools:
02.02 – Boat Designers: (Naval Architects, Boat Plans, Kits,+).
02.03 – Statutes & Standards: (Vessel & Equipment Requirements,+).
02.03.01 – Marine Laws & Regulations: (CAN, GBR, USA,+).
02.03.02 – Industry Standards: (ISO, IMO, ABYC, NFPA,+).
02.03.03 – Classification Societies: (ABS, LR, DNV<GL, BV, RINA,+).
02.04 – Boat Building & Refitting Tools,+: (Vendors, Specs, Manuals, Recalls,+).
02.05 – Boat Materials: (Qualities, Vendors, Specs, Manuals, Recalls,+).
02.05.01 – Wood: (Species, Rot; Carvel, Clinker/Lapstrake, Veneer/Plywood, Cold Molded,+).
02.05.02 – Metal: Iron, Steel, Aluminum, Copper,+. (Strengths, Weaknesses, Corrosion,+).
02.05.03 – Ferrocement: (Strengths, Weaknesses, Corrosion,+).
02.05.04 – FRP & GRP Composites: Fiberglass, Carbon Fiber,+. (Strengths, Weaknesses,+).
02.06 – Boat Equipment: (Vendors, Specs, Manuals, Reviews, Recalls,+).
02.06.01 – Steering & Thrusters: (Mechanical, Electric, Hydraulic,+).
02.06.02 – Stabilizers & Trim Plates: (Mechanical, Electric, Hydraulic,+).
02.06.03 – Dewatering Devices: (Bailers, Bilge Pumps,+).
02.06.04 – Galvanic Corrosion Protection: (Active, Anodes,+).
02.06.05 – Hull Penetrations & Openings: (Thru-Hulls, Scuttles, Skylights, Hatches,+).
02.06.06 – Deck Hardware & Equipment: (Cleats, Railings,+).
02.06.06.01 – Ground Tackle: (Anchors, Rodes, Chocks, Winches,+).
02.06.06.02 – Commercial Fishing Gear: (Pots, Nets, Lines, Winches, Reels, Rods,+).
02.06.07 – Rigging: (Rig Types, Standing Rigging, Running Rigging, Vendors, Riggers,+).
02.06.07.01 – Sails: (Sail Types, Aerodynamics, Vendors, Sailmakers,+).
02.06.08 – Propulsion Machinery: (Types, Configurations, Features, Control Systems,+).
02.06.08.01 – Engines: (Systems, Manufacturers, Marinizers, Resellers,+).
02.06.08.01.01 – Engine Mechanical (Pistons, Rods, Crankshafts, Cylinders, Heads, Valves,+).
02.06.08.01.02 – Engine Lubrication (Splash, Forced, Oil, Filtration, Additives, Oil Analysis,+).
02.06.08.01.03 – Engine Aspiration & Fuel (Natural & Charged; Gasoline, Diesel, Bunker, NG,+).
02.06.08.01.04 – Engine Electrical (Cranking, Charging, Ignition, Instrumentation, Electronics,+).
02.06.08.01.05 – Engine Cooling (Air, Liquid: Raw Water, Fresh Water w/Keel Coolers, Heat Exchangers,+).
02.06.08.01.06 – Engine Exhaust (Dry, Wet w/Separators,+).
02.06.08.01.07 – Engine Mounting (Hard, Soft,+).
02.06.08.02 – Engine-to-Marine Gear Interfaces: (SAE Specs, Damper Plates, Jackshafts,+).
02.06.08.03 – Marine Gears: (Reversing, Reduction; Mechanical, Hydraulic).
02.06.08.04 – Shafting: (Propshafts, Couplings, Seals, Bearings, Struts, Keys, Nuts,+).
02.06.08.05 – Propellers: (Types, Materials,+).
02.06.09 – Electrical Systems: DC & AC (Direct Current: Batteries, Solar,+; Alternating Current,+).
02.06.09.01 – Auxiliary Generators: (Diesel, Gasoline, Natural Gas,+).
02.06.09.02 – DC-to-AC Inverters:
02.06.10 – Navigation & Communication Systems: (Charting, GPS. Radar; WX, SAT, HF, VHF,+)
02.06.11 – Safety Equipment: (Life Rafts, PFDs, Firefighting Eq., Alarms, Medical Kits,+).
02.06.12 – Domestic Systems:
02.06.12.01 – LPG & CNG Systems:
02.06.12.02 – Cabin Heating & Cooling: (Heaters, Heat Pumps; Air Conditioning,+)
02.06.12.03 – Galley Appliances: (Refrigeration, Galley Stoves,+).
02.06.12.04 – Water & Waste Systems: (Tanks, Water Makers, Pumps, Plumbing,+)
02.06.12.05 – Trash Disposal:
02.06.12.06 – Furnishings: (Cabinetry, furniture, Coverings, Entertainment, Weather,+).
02.06.13 – Personal Equipment:
02.06.13.01 – Diving: (Commercial & Sport).
02.06.13.02 – Fishing: (Sport).
02.06.13.03 – Racing: (Sail, Offshore Power, Powerboat, Hydroplane,+).
02.06.13.04 – Sailing: (Foul Weather Gear, Safety Harnesses,+).
02.06.13.05 – Watersports: (Surfing, Skiing, Boarding, Tubing,+).
02.06.14 – Boat Tenders:
02.06.15 – Boat Trailers: (Bunk, Roller,+)
02.07 – Marine Suppliers: (CAN, GBR, USA,+). (National Chains, Local,+)
02.08 – Boat Builders: (A∼Z) (w/Vessel Types, Locale & Years Active)
02.09 – Boat Refitters: (CAN, GBR, USA,+) (Shipyards, Boatyards, Riggers, Repair Shops,+).
02.10 – Boat Building, Outfitting, Refitting & Repair Schools: (Incl. DIY).
02.11 – DIY Boat Building, Outfitting, Refitting & Repair: (Maintenance‚ Troubleshooting‚+).
15 – DO-IT-YOURSELF (DIY):
15.01 – DIY Boat Building, Outfitting, Refitting & Repair: (Incl. Maintenance & Fault Finding).
15.02 – DIY Boat Sales: (Buyers & Sellers).
15.03 – DIY Boat Inspections: (Pre-Survey, Pre-Purchase, Pre-Sale, Pre-Voyage, Sea Trials,+).
15.04 – DIY Schools & Classes: (Boat Building, Refitting, Inspection, Troubleshooting, Repair,+).
16 – MEDIA w/Creator Directory: (Authors, Editors, Publishers,+) + Academy eLibrary.
16.01 – Documentation: (Catalogs, Ads, SpecSheets, Manuals, TechVids, Bulletins, Recalls‚+).
16.02 – Books: (Bound, eBooks,+).
16.03 – Magazines: (Incl. Back Issues, Articles,+).
16.04 – Videos: (How-to-Tutorials, Documentaries, Travelogues, Movies,+).
16.05 – Websites: (Incl. Articles, Forum Posts, Tech Tips, Tech Notes, Social Media,+).
EVERYTHING ON THIS PAGE OK?
If there is anything on this webpage that needs fixing, please let us know via email To:
Editor♥EverythingAboutBoats.org (Replace "♥" with "@")
THIS ARTICLE IS STILL EVOLVING!
The page may contain rough drafts that include raw source materials.

Visit our FEATURED ARTICLES Home Page
to see examples of our website's comprehensive contents!
Thanks to our amazing contributors for the steady flow of articles, and to our dedicated all-volunteer staff who sort, polish and format them, everyday we get a little bit closer to our goal of
Everything About Boats. If you would like to submit an article,
See Submitting Articles.
— TOP 20 MOST POPULAR ARTICLES —
Detroit Diesel 8.2 Liter “Fuel Pincher” V8 Engine
Cummins V-555 & VT-555 “Triple-Nickel” V8 Diesel Engine
Lehman 120 (6D380) Diesel Engine (Ford 2704C & 2715E)
Ford Industrial Power Products Diesel Engines
How to Identify Ford Diesel Engines
Ford 2715E Diesel Engine
Lehman Mfg. Co.
Perkins Engines
Universal Atomic 4
Sears Boat Motors: Motorgo, Waterwitch, Elgin, etc.
Chrysler & Force Outboards
Eska Outboard Motors
Allison Transmission
ZF Friedrichshafen AG
Marine Surveyors by Country
American Marine Ltd (Grand Banks)
Boat Inspection (Types of Marine Surveys)
Boat Builders: (A∼Z) (w/Vessel Types, Locale & Years Active)
USCG NVIC 07-95 Guidance on Inspection, Repair and Maintenance of Wooden Hulls
American Boat and Yacht Counsel (ABYC)
Layout of the EverythingAboutBoats.org Website's Pages
— Types of Webpages —
This website consists almost entirely of 3 types of webpages as follows:
- TOPIC PAGES (See Main Topic Pages listed on Website Contents or the Right Sidebar) ⇒
- EXAMPLE: – WEBSITE CONTENTS w/Links to MAIN TOPIC & Subtopic Pages:
- EXAMPLE: 02 – BOAT BUILDING, OUTFITTING, REFITTING & REPAIR:
- EXAMPLE: 02.06 – Boat Equipment:
- EXAMPLE: 02.06.08 – Propulsion Machinery:
- EXAMPLE: 02.06.08.01 – Engines w/Directory of Vendors.
- VENDOR PAGES (Vendors of Products, Services, Events,+, Destinations & Media Creators)
- EXAMPLE: 02.06.08.01 – ^ Ford Industrial Power Products Diesel Engines.
- PRODUCT PAGES (Equipment, Events, Media: pDoc, Books, Magazines, Videos, Websites,+)
- EXAMPLE: 02.06.08.01 – ^ ^ Ford 2715E Diesel Engine.
- EXAMPLE: 02.06.08.01 – ^ ^ Ford OpManual - 2710 Range Diesel Engines.
- EXAMPLE: 02.06.08.01 – ^ ^ Ford Service Manual - 2700 & 2710 Range Diesel Engines.
Clickable Links that lead to other webpages appear in Blue Text and usually open in a new window.
Links in the Right Sidebar and most directories open in the current window, not a new window.
Note in the examples above that these pages form a natural hierarchy.
The unnumbered "^" pages are listed alphabetically in most tables.
Media Titles in tables are distinguished by their smaller font size.
Media (Books, Magazines, Videos, Articles,+) are treated as Products.
Vendors' Product Documentation (pDoc) are considered Media.
Destinations & Media Creators are treated as Vendors.
All Website Pages are optimized for viewing on
full-width disktop computer monitors,
but can be viewed on phones.
— Contents of Webpages —
Website Pages typically contain the following Sections:
- PATH (Shows the chain of EAB pages w/links that lead to the page being viewed).
- EXAMPLE:
PATH: HOME, WEBSITE INDEX, WEBSITE CONTENTS » ∨
∧ BOAT BUILDING & REPAIR » Boat Equipment » Propulsion » Engines » ∨∨
∧∧ Ford, Ebro, American Diesel, AmMarine, Barr, Beta, Bomac, Bowman, Couach,
Lees, Lehman, Mermaid, Parsons, Renault, Sabre, Thornycroft, Wortham Blake »
∧ DO-IT-YOURSELF » DIY Boat Building & Repair » DIY Schools & Classes »
∧ MEDIA w/Creator Directory » Documentation, Books, Magazines, Videos, Websites » - (The "»" right pointing Guillemet symbol shows the chain through the page links.)
- (The "," comma between page links in the chain indicates pages are not subordinate, but are instead at the same level. See engine brands in the example above.)
- (The "∨", "∨∨", "∨∨∨",+ symbols indicate that the path line continues with whatever follows the "∧", "∧∧", "∧∧∧",+ symbols respectively. "∧" Precedes each MAIN TOPIC Page.)
- EXAMPLE:
- PAGE CONTENTS (Table of Contents with links to each main section on the page).
- PAGE BODY (The type of page determines the contents of its body as follows:).
- TOPIC PAGES (Topic Treatment: Introduction, Overview, Background, Details,+).
- (Many Topic Pages contain Directories of Vendors with Links).
- (Most Directory Listings are Alphabetical and/or by Locale).
- VENDOR PAGES (Vendor's Profile, Contact Information, Products, Services,+).
- (Manufacturers, Resellers, Refitters, Yards, Surveyors, Clubs, Schools, Authors,+).
- (Boating & Travel Destinations are treated as Vendors on their own Vendor Pages).
- PRODUCT PAGES (Product Features, Vendor Links, Specifications, Documentation,+).
- (Media created by a vendor is often treated as a Product on its own Product Page).
- (Boating & Travel Events are often treated as Products on their own Product Pages).
- TOPIC PAGES (Topic Treatment: Introduction, Overview, Background, Details,+).
- RELATED RESOURCES (Topics, Vendors, Products, Media: Books, Websites,+ with Links).
- PAGE TAIL Contains the following Anchors Aweigh Academy & EAB Website Features:
- The Anchors Aweigh Academy's EverythingAboutBoats.org Header.
- A link to our Featured Articles EAB Home Page.
- Top 20 Most Popular Articles. (The section that appears right above ⇑ this section).
- Layout of the EverythingAboutBoats.org Website's Pages. (This very section ⇐).
- Topics of Webpages. (The very next section below ⇓).
- What we have accomplished so far.
- Members must Sign-In to gain full access to Expanded Pages & Programs.
- Sign-Up (if not already a member).
- Public Comments (about the website & about this page).
- RIGHT SIDEBAR (Website Contents menu with links to Main Topic & Subtopic pages).
(On some smart phones, the Right Sidebar may appear at the bottom of the webpage)
— Topics of Webpages —
Website Pages are categorized under the following 16 MAIN TOPICS:
The MAIN TOPICS follow a natural progression from conception of the vessel thru its
building, marketing, survey, financing, insuring, transport, moorage, use and upkeep.
The MAIN TOPICS (all Caps) below are followed by their Main Subtopics with Links.
00 – HOME: CONTENTS, ABOUT EAB: Contact EAB, Abbreviations & Symbols, FAQ, GLOSSARY, ADs,+.
01 – ABOUT BOATS w/Museum Directory: Early History, Recent History, Modern Vessel Types,+.
02 – BOAT BUILDING, OUTFITTING, REFITTING & REPAIR: Materials, Equipment, Builders,+.
03 – BOAT MARKETING: Boat Shows, Dealers & Brokers, Importing & Exporting, Auctions & Sales,+.
04 – BOAT INSPECTION: Types of Marine Surveys, Marine Surveyors, Schools, DIY Inspections,+.
05 – BOAT TITLES & VESSEL REGISTRY: Boat Title & Registration, Vessel Registry, Title Co's,+.
06 – BOAT FINANCING: Conventional (Banks, Credit Unions,+), Unconventional (Creative),+.
07 – BOAT INSURANCE: Maritime & Recreational: Coverage, Carriers, Agents,+., Claim Processing,+.
08 – BOAT TRANSPORT: By Sea (Piggyback, Delivery Skippers & Crews, & Towing), Over-Land,+.
09 – BOAT HAULING & LAUNCHING: Drydocks, Ways, Lifts, Cranes & Hoists, Launch Ramps,+.
10 – BOAT MOORAGE & STORAGE: Builders, Anchorages, Marinas, Yards, Racks & Stacks,+.
11 – BOATING ORGANIZATIONS: Yacht Clubs & Sailing Clubs, Paddling Clubs, Boat Owners,+.
12 – BOATING & TRAVEL: Events, Destinations, Boat Rentals & Charters, Cruises, Voyages,+.
13 – BOATING & MARITIME EDUCATION: Recreational Seamanship, Ship's Master & Crew,+.
14 – MARINE LAWS & REGULATIONS: International & National Laws‚ Lawyers‚ Investigators‚+.
15 – DO-IT-YOURSELF: DIY Boat Building & Repair, DIY Boat Sales, DIY Boat Surveys, DIY Classes,+.
16 – MEDIA w/Creator Directory + Academy eLibrary: pDocs, Books, Magazines, Videos, Websites,+.
The above MAIN TOPICS and a more detailed listing of Subtopics
can be found on the Website Contents page and on the Right Sidebar.
An Alphabetical Index of the website can be accessed at Website Index.
What we have accomplished so far.
Anchors Aweigh Academy and its EverythingAboutBoats.org website.
- Published over 50,000 website pages about boats and boating, bringing us closer to reaching our goal of becoming "The ultimate reference resource about boats and ships for everyone from the beginning recreational boater to the seasoned professional mariner!"
- Published over 300 website main topic webpages, many with full articles on the topic. See our Website Contents or the Right Sidebar for the listing of the main topic pages.
- Published over 9,000 marine vendor webpages, all with their contact information, most with a description of their products and services, many with product documentation, specifications and independent reviews. (incl.: Boat designers, boat building tools, material and equipment manufacturers and suppliers, boat builders and dealers, yacht brokers, marine surveyors, boat insurers, boat transporters, skippers and crews, boatyards and marinas, yacht clubs, boat rentals and yacht charters, boating, seamanship and maritime schools, marine law attorneys and expert witnesses, boat refitters and repairers, book authors, magazine publishers, video producers, and website creators)
- Acquired over 120,000 pages of product documentation including Catalogs, Brochures, SpecSheets, Pictures, Serial Number Guides, Installation Manuals, OpManuals, Parts Catalogs, Parts Bulletins, Shop Manuals, Wiring Diagrams, Service Bulletins, and Recalls. And have made all viewable to Academy Members through our EAB website eLibrary.
- Acquired over 1,200 books and magazine back issues in our academy library and so far have made over 700 viewable to Academy Members through our EAB website eLibrary.
- Published over 500 DIY How-To articles about boat design, construction, inspection, operation, maintenance, troubleshooting and repair. We are working hard to do more.
We are currently formatting and polishing the Anchors Aweigh Academy online and hands-on courses. Our Marine Surveying course has proven to be excellent for both the beginner and the seasoned surveyor, and especially helpful to the Do-It-Yourselfer.
Current Academy Members must SIGN IN to gain FULL access to this
website including expanded pages and valuable Academy programs
like our Academy eLibrary and our Ask-An-Expert Program!
If your membership has expired, CLICK HERE to Renew.
IF YOU ARE NOT YET AN ANCHORS AWEIGH ACADEMY MEMBER,
CLICK HERE to discover how you can become a Member and gain FULL access to
thousands of expanded pages and articles, and dozens of excellent programs
WITH JUST A SMALL DONATION!
Comments for Public Viewing
Submit any comments for public viewing via email To: Comments♥EverthingAboutBoats.org (Replace "♥" with "@")
Please remember to put this webpage's title in the subject line of your email.
All comments are moderated before they appear on this page. See Comment Rules.
General Comments About the Website
FROM Donald: "This is an awesome website. I found the information that I needed right away from one of the over 20,000 free articles that you provide as a public service. I'm surprised that so much if this site is free. But I still signed up so I could access the thousands of expanded pages, interesting articles, and dozens of valuable programs! The member's library of books, magazines and videos that I can view online is really terrific! I understand that you and your staff are all unpaid volunteers. Please keep up the good work. And I commend you for your plans to add another 10,000 free informative articles over the next year. I'm thrilled to support you in this endeavor with my small membership donation. Thanks again for all your hard work."
FROM Huey: "I agree with my Uncle, I too have found the articles to be very enlightening. They say that it will take about 100,000 articles to cover the full scope that they have envisioned for the website. They have over 20,000 articles so far and that's doing pretty well, but it could take several years to get the rest. I also noticed that many of the Main Topic Pages and some of the article pages are still in the rough draft stage. I guess that they will fill in as they can get volunteers to work on them. But what I can't figure out is why anyone would spend the time writing informative in depth articles just to give away free to this website for publication? What's in it for them?"
FROM Dewey: "Well Huey, to me It looks like most of the articles on this website are written by very informed people, like boating instructors, boat designers, boat builders, riggers, electricians, fitters, marine repair technicians and marine surveyors. Writing such articles helps establish them as knowledgeable professionals. After all, this website was originally created by a school for marine technicians and marine surveyors. The website is growing in content every day. They even had to move to a bigger, more powerful server because the website's traffic has been growing exponentially."
FROM Louie: "I agree with everyone above. This site is quickly becoming the ultimate reference resource about every aspect of boats and ships for everyone from the beginning recreational boater to the seasoned professional mariner. I use the topic pages on the right sidebar to browse around the website. It's like a Junior Woodchucks' Guidebook for Boaters. Their Members' Library of over 300 popular and obscure books and over 200 magazine back issues that can be viewed online is fabulous. The Academy's magazine is especially informative. On top of that, there is the "Ask-An-Expert program for members where you can get an expert's answer to any of your boat questions. And a whole years membership is only $25. What a deal! I really love being part of this "Everything About Boats" community and help provide thousands of helpful articles free to the public. I think that I'll sit down right now and write an article about my experiences boating with my uncle."
FROM Scrooge: "You rave about this website like it was the best thing since sliced bread. Well, I think it stinks. Sure, it has a lot of good information for boaters, and they're adding more every day, but it will probably never be finished. Furthermore, I don't even own a boat. And I wouldn't have a boat even if someone gave me one. Boats are a waste of money and time and energy and money! They're just a hole in the water you pour money into. If you gave me a boat, I'd sell it quicker then you could say Baggywrinkle. Then I'd lock up the cash with all my other money so I could keep my eye on it and count it every day. Bah humbug."
FROM Daisy: "I'm just so glad that Donald got the boat so we and the boys could enjoy boating — together. And of course all of the girls, April, May, and June, love to be on the water too, especially when that is where the boys are. Oh poor Scrooge, boating is more fun then you could possibly imagine."
FROM Scrooge: "After seeing how much fun you all have on the water together, I regret that I didn't have that much fun when I was young. I've had a change of heart, and I'm giving each of you a Lifetime Academy Membership."
FROM Editor: "For those of you that have stayed with us this far, many thanks, and we hope that you found this little narrative informative. Your faithful support inspires us to keep working on this phenomenal website. We know that we have a lot more to do. Ultimately, we hope that we can help you enjoy the wonder filled world of boating as much as we do. We are all waiting to see what you have to say about this webpage article. Submit any comments via email To: Comments♥EverythingAboutBoats.org (Replace "♥" with "@"). Be sure to include this page's title in the subject line. Also, your corrections, updates, additions and suggestions are welcomed. Please submit them via email To: Editor♥EverythingAboutBoats.org (Replace "♥" with "@"). It has been truly amazing to see what we have been able to accomplished when we've worked together. Thanks to all those that have donated their valuable time and energy, and a special THANK YOU to all that have supported this cause with their membership donations."
Comments About This Particular Page
FROM ΞNameΞ: “Be_the_next_to_comment_about_this_page.” {230316}